[latexpage]
Illustrative Example
This page provides an example of how a user would input information into the BRCAlive Tool, followed by a detailed description of the underlying cancer risk calculations. The BRCAlive Tool provides breast and ovarian cancer probability estimates, by year, from age 20 to 90.
Example: Jane Doe is currently 38 years old, with a known deleterious BRCA mutation (c.3178) as shown in her genetic test report. After entering this information, the model estimates her future lifetime risks cancer in the absence of any preventive surgery.
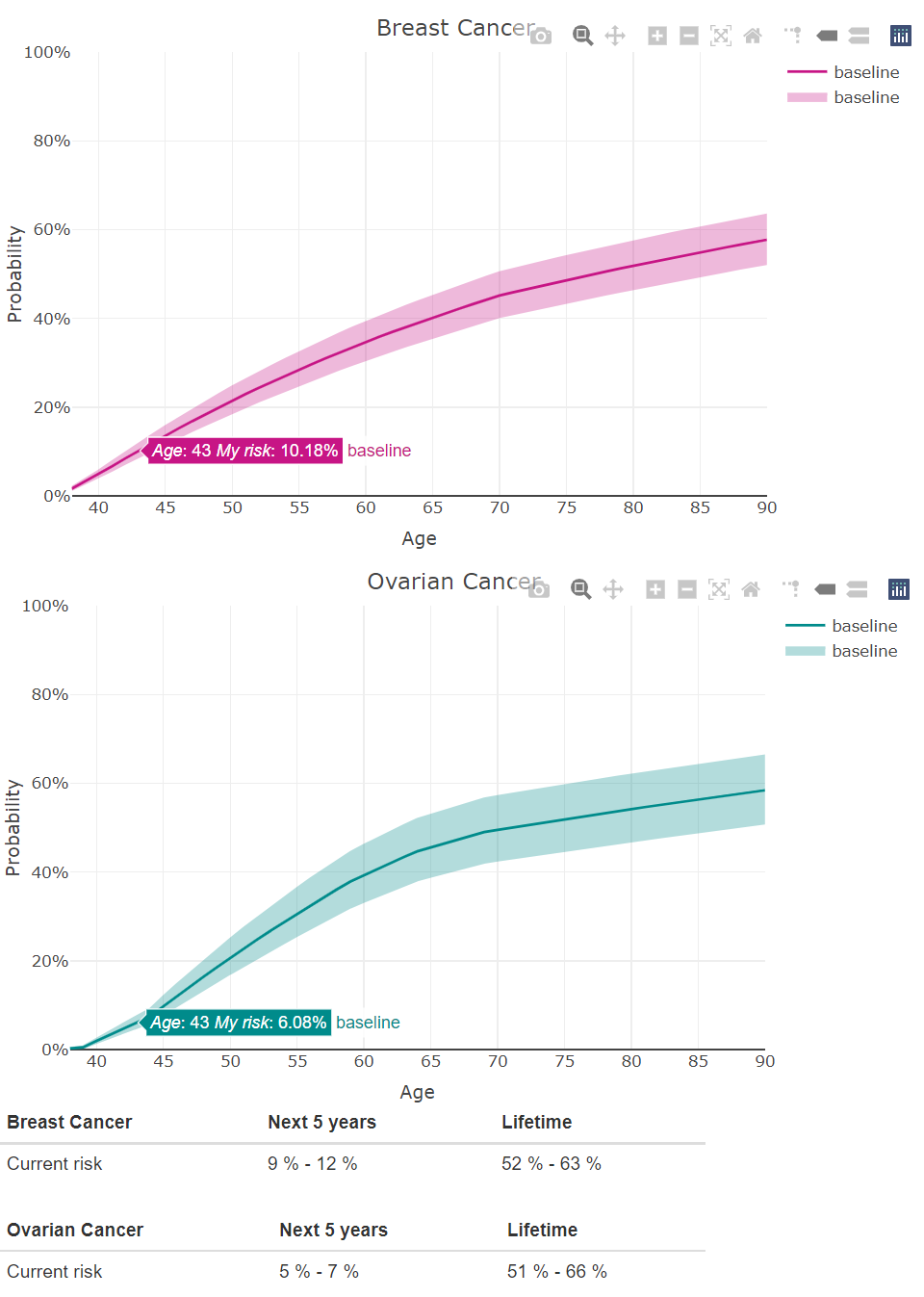
Jane’s estimated risk of breast cancer over the next 5 years (by age 43) is 10%, with a range of 9-12%. In other words, our best guess of her cancer risk is 10%, but we are pretty confident that the risk falls between 9% and 12%. Over her lifetime (by age 90), her risk of breast cancer is 52-63%, assuming she does not undergo any preventive surgery. The tool allows her to see the estimated risk for any age, just by moving her mouse over the selected age.
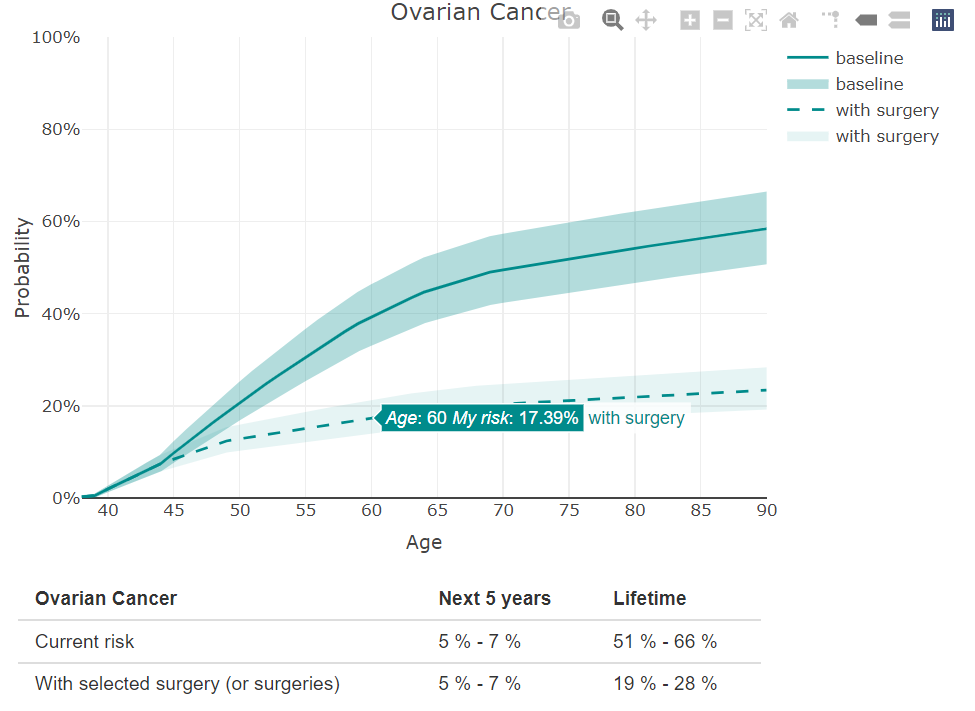
Jane’s estimated risk of ovarian cancer over the next 5 years (by age 43) is 6%, with a range of 5-7%, assuming that she does not undergo any preventive surgery. Her lifetime risk (by age 90) of ovarian cancer is 51-66%. As with breast cancer, she can see the probability for any age by just moving the mouse.
Breast Surgery
To account for the benefits of a prophylactic bilateral mastectomy (BM) on reducing cancer risk, a user selects whether she has previously undergone this surgery (either due to a breast cancer diagnosis, or as a prophylactic surgery), and whether she is considering it in the future.

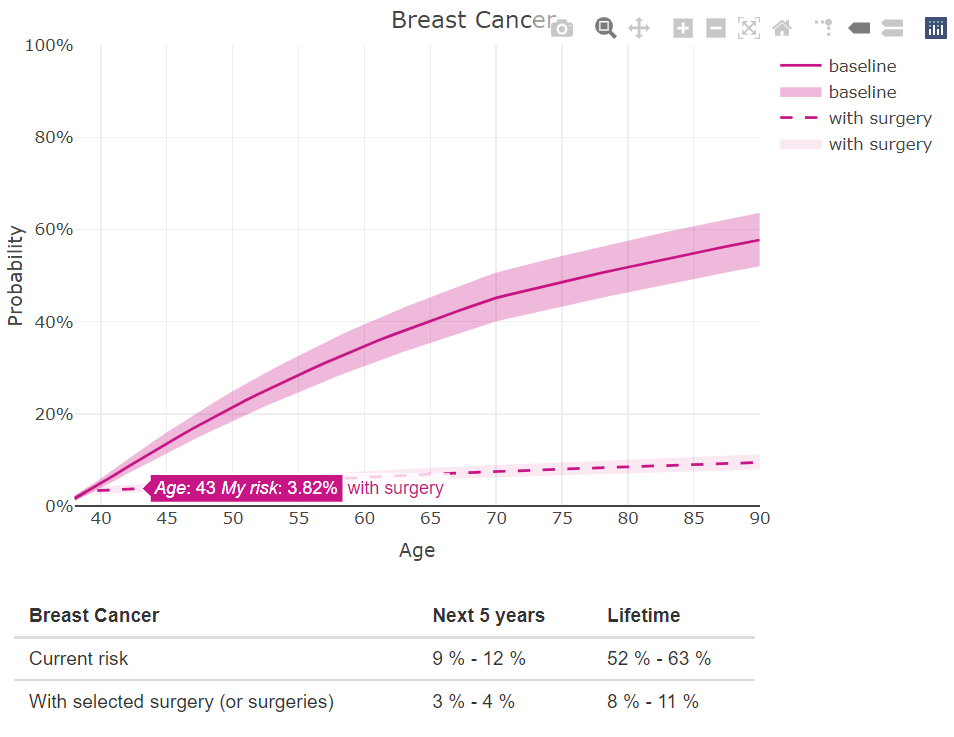
The graph adds a second dashed line to compare the baseline risk of breast cancer (without surgery) versus the estimated risk following the surgery. The user can easily move the slider below the graph to select different ages to undergo surgery.
Jane is contemplating having a prophylactic bilateral mastectomy (BM) in 2 years. She moves the slider to select age 40, and we see that her 5-year risk of breast cancer drops from 9-12% (with no surgery) to 3-4% (with BM at age 40). There is still some residual risk even with the surgery, and this also includes the risk over the next 2 years before the surgery. By age 90, her risk of breast cancer drops from 52-63% to 8-11%. Her breast cancer risk could be further reduced if she also removes her ovaries, as described next.
Ovarian and Fallopian Tube Surgery
The model accounts for the cancer risk-reducing benefits of a prophylactic bilateral oophorectomy (BO), the surgical removal of both ovaries (and fallopian tubes if applicable). The user selects whether she has previously undergone this surgery (either due to an ovarian cancer diagnosis, or as a prophylactic surgery), and whether she is considering it in the future.
The model also accounts for just a bilateral salpingectomy (BS), the surgical removal of just the fallopian tubes. The user can select one or both surgeries, or stage them at different ages.
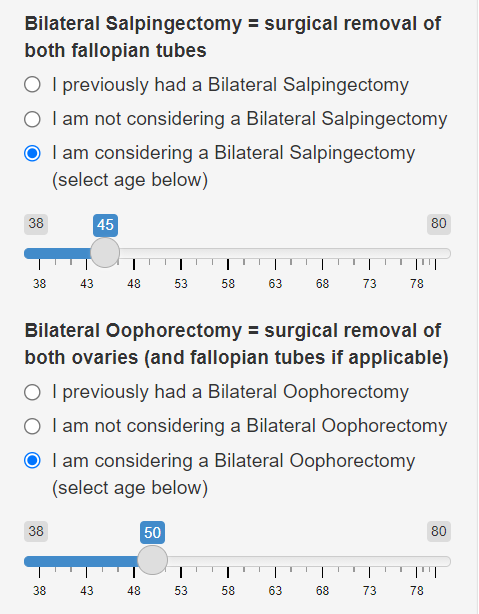
In addition to having a bilateral mastectomy at age 40, Jane is considering having her fallopian tubes removed at age 45 (a bilateral salpingectomy) and removing her ovaries (a bilateral oophorectomy) at age 50. She moves the sliders to select the different ages, and we see that her 5-year risk of ovarian cancer does not change because the first surgery is 7 years away. Her ovarian cancer risk by age 60 drops from 39% (with no surgery) to 17% (with BS at age 45 and BO at age 50). Moreover, her breast cancer risk by age 90 drops even further to 7-9%, since she is opting to undergo both a BM and BO. Both cancer risks could be slightly further reduced if she underwent the surgeries earlier.
Technical Details
The following sections contain detailed descriptions of the BRCAlive Tool’s cancer risk calculations and related published studies. This information is provided for research and educational purposes.
Baseline Breast Cancer Risk
We first calculate the baseline risk of breast cancer based on a prospective cohort study of women involving 6,036 BRCA1 and 3,820 BRCA2 carriers (Kuchenbaecker et al, 2017). These risks are then adjusted based on a woman’s specific mutation location, as described later.
Define the following terms:
Based on the Kuchenbaecker et al (2017) study, we assume the following annual age-adjusted rate of developing breast cancer for BRCA1 and BRCA2 carriers: $$\lambda_1^{BC}(t)=\begin{cases} 0.0054& \text{ for } t=21,…,30 \\ 0.0209& \text{ for } t=31,…,40 \\ 0.0244 & \text{ for } t=41,…,50 \\ 0.0234& \text{ for } t=51,…,60 \\ 0.0220& \text{ for } t=61,…,70 \\ 0.0165& \text{ for } t=71,…,90 \end{cases}$$ $$\lambda_2^{BC}(t)=\begin{cases} 0.0038& \text{ for } t=21,…,30 \\ 0.0079& \text{ for } t=31,…,40 \\ 0.0213 & \text{ for } t=41,…,50 \\ 0.0233 & \text{ for } t=51,…,60 \\ 0.0226& \text{ for } t=61,…,70 \\ 0.0218& \text{ for } t=71,…,90 \end{cases}$$
We use these estimates to calculate the aggregate risk of developing breast cancer by a certain age, for our simulated cohort of 10,000 women.


Source: Kuchenbaecker KB et al. Risks of Breast, Ovarian, and Contralateral Breast Cancer for BRCA1 and BRCA2 Mutation Carriers. JAMA. 2017;317(23):2402-2416. doi:10.1001/jama.2017.7112
Baseline Ovarian Cancer Risk
We first calculate the baseline risk of ovarian cancer based on a registry of women involving 4,316 BRCA1 and 1,373 BRCA2 carriers (Kotsopoulos et al, 2018). As with breast cancer, these risks are then adjusted based on a woman’s specific mutation location, as described later.
Define the following terms:
Based on the Kotsopoulos et al (2018) study, we assume the following annual age-adjusted rate of developing ovarian cancer for BRCA1 and BRCA2 carriers: $$\lambda_1^{OC}(t)=\begin{cases} 0.0000& \text{ for } t= 21,…,29\\ 0.0011& \text{ for } t= 30,…,34\\ 0.0021& \text{ for } t= 35,…,39\\ 0.0117& \text{ for } t= 40,…,44\\ 0.0208& \text{ for } t= 45,…,49\\ 0.0216 & \text{ for } t= 50,…,54\\ 0.0225 & \text{ for } t= 55,…,59\\ 0.0189& \text{ for } t= 60,…,64\\ 0.0133& \text{ for } t= 65,…,69\\ 0.0079& \text{ for } t= 70,…,90 \end{cases}$$ $$\lambda_2^{OC}(t)=\begin{cases} 0.0000& \text{ for } t= 21,…,29\\ 0.0000& \text{ for } t= 30,…,34\\ 0.0000& \text{ for } t= 35,…,39\\ 0.0011& \text{ for } t= 40,…,44\\ 0.0013& \text{ for } t= 45,…,49\\ 0.0030 & \text{ for } t= 50,…,54\\ 0.0075 & \text{ for } t= 55,…,59\\ 0.0157& \text{ for } t= 60,…,64\\ 0.0105& \text{ for } t= 65,…,69\\ 0.0037& \text{ for } t= 70,…,90 \end{cases}$$
We use these estimates to calculate the aggregate risk of developing ovarian cancer by a certain age, for our simulated cohort of 10,000 women.
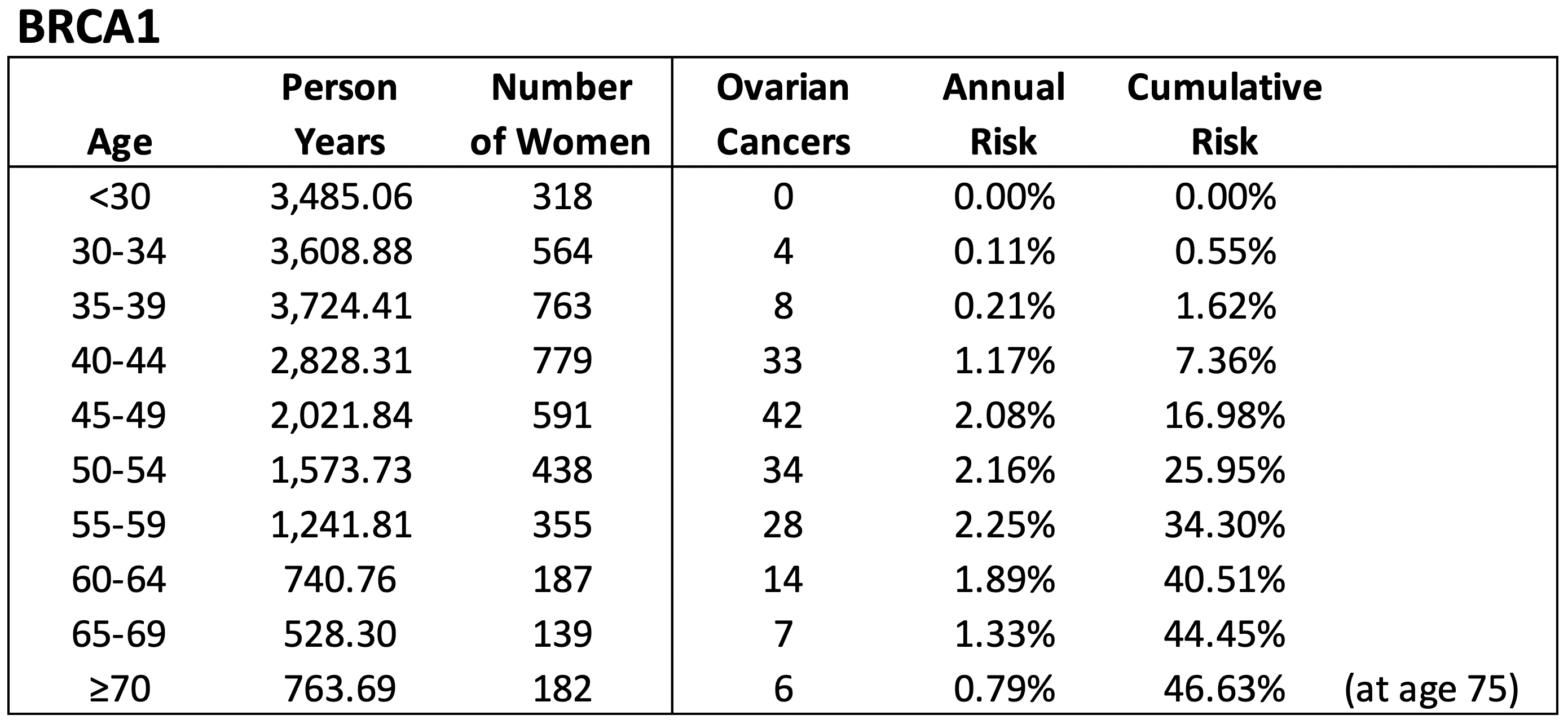
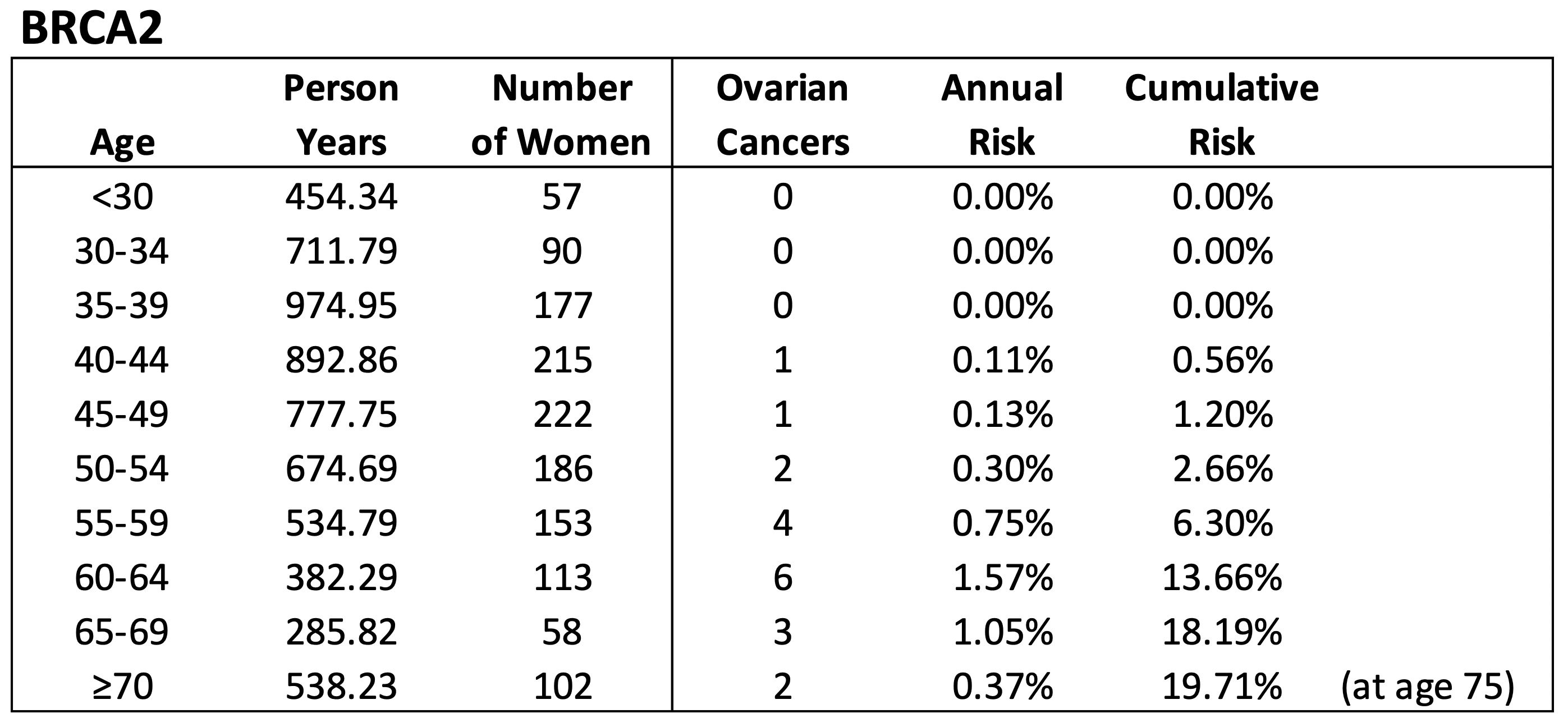
Source: Kotsopoulos J et al. Age-Specific Ovarian Cancer Risks among Women with a BRCA1 or BRCA2 Mutation. Gynecologic Oncology. 2018;150(1):85-91. doi:10.1016/j.ygyno.2018.05.011
Mutation Location
We adjust the baseline risk of developing breast and/or ovarian cancer based on the location of a woman’s BRCA genetic mutation, using data from an observational study involving 19,581 BRCA1 and 11,900 BRCA2 carriers (Rebbeck et al, 2015).
BRCA1 mutations are categorized into 30 bins, including 2 bins corresponding to the Ashkenazi Jewish founder mutations c.68_69delAG (bin #1) and c.5266dupC (bin #29). BRCA2 mutations are categorized into 19 bins, including the Ashkenazi Jewish founder mutation c.5946delT (bin #11).
For each mutation bin, Rebbeck et al (2015) estimate the hazard ratio of developing breast or ovarian cancer. A hazard ratio of 1.0 assumes that the risk of cancer is no different than the baseline estimate for all BRCA1 or BRCA2 carriers. A hazard ratio greater than 1.0 implies a higher risk; a value less than 1.0 implies a lower risk. For example, a hazard ratio of 2.0 means that cancer risk is twice as high as the baseline rate.
Define the following terms:
We assume that the mutation-specific hazard ratio is constant across all ages. The authors also provide 95% confidence intervals (CI), which represent an upper and lower range of values where we are 95% confident the true hazard ratio falls between, based on the available data. Note that bins that include more women, such as BRCA1 bin #2, have narrower confidence intervals, suggesting that we have a better estimate of the hazard ratio associated with mutations in this bin.
We multiply this hazard ratio by the baseline cancer risk to obtain the mutation-specific adjusted risk of breast cancer:
We calculate the mutation-specific adjusted risk ovarian cancer in a similar manner, for both BRCA1 and BRCA2 mutation carriers.

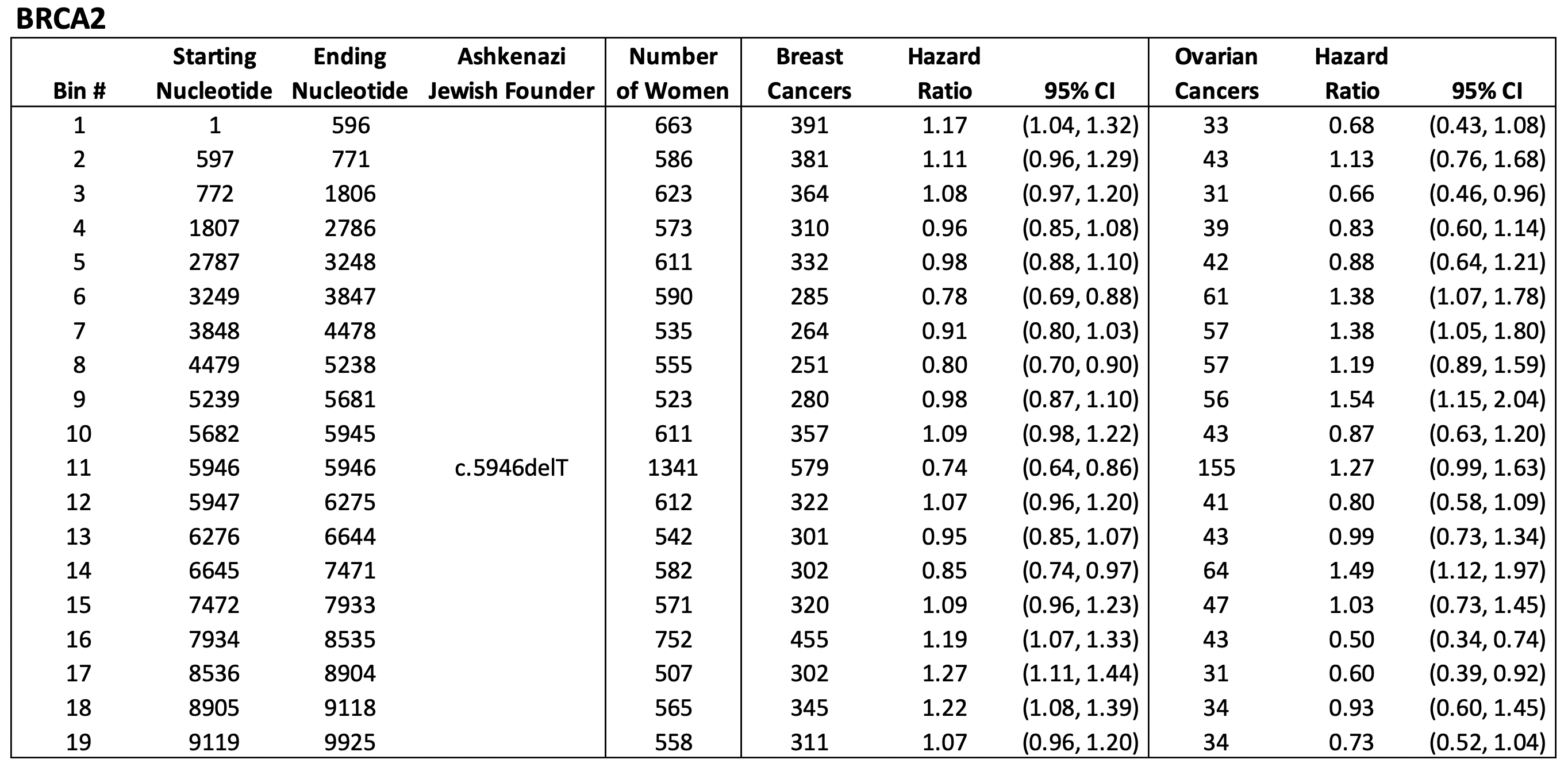
Source: Rebbeck TR et al. Association of Type and Location of BRCA1 and BRCA2 Mutations with Risk of Breast and Ovarian Cancer. JAMA. 2015;313(13):1347-1361. doi:10.1001/jama.2014.5985
Surgery Risk-Reduction
For a detailed review of studies demonstrating the benefits of prophylactic (risk-reducing) surgery on breast and ovarian cancer in BRCA carriers, please see:
Hartmann LC, Lindor NM. The Role of Risk-Reducing Surgery in Hereditary Breast and Ovarian Cancer. New England Journal of Medicine. 2016;374(5):454-468. doi:10.1056/NEJMra1503523
Prophylactic Bilateral Mastectomy (BM)
Many studies have demonstrated a substantial reduction in breast cancer risk following a prophylactic bilateral mastectomy (BM), which is the surgical removal of both breasts. In one study of 639 women with a strong family history of breast cancer, the authors estimated a 92% reduction (HR = 0.08) in breast cancer risk following a BM (Hartmann et al, 1999). Note, this analysis occurred before the BRCA genetic mutation was discovered; hence we are unable to know whether women in this study have BRCA mutations. A hazard ratio of 1.0 indicates no change in breast cancer risk; values less than 1.0 indicate a decreased risk; values greater than 1.0 indicate an increased risk.
In a second study involving 483 women with known BRCA mutations, the authors estimate a 91% reduction (HR = 0.09) in breast cancer risk following a BM. (Rebbeck et al, 2004).
For the BRCAlive Tool, we assume a 92% reduction (HR = 0.08) in our breast cancer risk calculations. $$\text{HR}_{PBM}^{BC}=0.08$$
The model further adjusts the age-specific and mutation-specific risk of breast cancer by the hazard ratio following a BM: $$\tilde{\lambda}_{1,j}^{BC}(t)=\text{HR}_{BM}^{BC}\times \bar{\lambda}_{1,j}^{BC}(t)$$
Prophylactic Bilateral Salpingo-Oophorectomy (BSO)
A prophylactic bilateral salpingo-oophorectomy (BSO) involves surgically removing both ovaries and fallopian tubes; it does not necessarily include a hysterectomy (removing the uterus). One study of 5,783 women with a BRCA1 or BRCA2 mutation found an 80% reduction (HR = 0.20) in the risk of ovarian, fallopian, or peritoneal cancer following a PBSO (Finch et al, 2014). $$\text{HR}_{BSO}^{OC}=0.20$$
A second important benefit of undergoing a BSO is a reduction in breast cancer risk, because of reduced hormone production, which can feed breast tumor growth. One study involving 869 BRCA1 and 501 BRCA2 carriers estimated breast cancer risk reduction following with a BSO (Domcheck et al, 2010). The authors estimated a 37% reduction (HR = 0.63) in breast cancer risk for BRCA1 carriers, and a 64% reduction (HR = 0.36) for BRCA2 carriers (Domcheck et al, 2010). $$\text{HR}_{BSO,1}^{BC}=0.63$$ $$\text{HR}_{BSO,2}^{BC}=0.36$$
One explanation as to why a BSO confers greater benefit (a lower HR) for BRCA2 carriers is because breast tumors in BRCA2 carriers tend to be hormone-receptor positive. In a different study of 4,325 BRCA1 and 2,568 BRCA2 carriers, 22% of breast tumors in BRCA1 carriers were estrogen receptor-positive, compared to 77% in BRCA2 carriers (Mavaddat et al, 2012). Additionally, 69% of tumors in BRCA1 carriers were “triple-negative” (estrogen receptor-negative, progesterone receptor-negative, HER2-negative), compared to only 16% of tumors in BRCA2 carriers. Following a BSO, the amount of estrogen produced in the body substantially drops, potentially explaining why a BSO reduces breast cancer risk more in BRCA2 carriers.
Undergoing both a BM and BSO offers the greatest reduction in breast cancer risk. Rebbeck et al (2004) estimate a 95% reduction (HR = 0.05) in breast cancer risk following both surgeries, which is what we assume in our simulation model. $$\text{HR}_{BM,BSO}^{BC}=0.05$$
Prophylactic Bilateral Salpingectomy (BS)
A prophylactic bilateral salpingectomy (BS) involves surgically removing only the fallopian tubes. Although we have less data on the effectiveness of a BS at preventing ovarian cancer in high-risk BRCA mutation carriers, one retrospective study of 3,319 BRCA carriers found a 57% reduction (HR = 0.43) in the risk of ovarian cancer following fallopian tube ligation (Antoniou et al, 2009). Some other studies found a more modest reduction of around 30%, on average, but most patients were not BRCA mutation carriers (Rice et al, 2021). We therefore assume a 57% reduction in ovarian cancer risk for both BRCA1 and BRCA2 carriers. Note, there is no reduction in breast cancer risk following only a BS because the ovaries are still intact. $$\text{HR}_{BS}^{OC}=0.43$$
Multiple Surgeries
The model calculates the overall adjusted risks of breast and ovarian cancer based on a user’s input regarding the timing of a BM, BS, and/or BSO. For the two gynecological surgeries, we assume that the risk reduction conferred by a BSO supersedes that of a BS only. For example, if a woman undergoes a BS at age 30 and subsequently has a bilateral oophorectomy at age 40, her annual risk for ovarian cancer will be multiplied by 0.43 from age 30 to 40, and by 0.20 from age 40 onwards.
References
- Domchek SM, Friebel TM, Singer CF, Evans DG, Lynch HT, Isaacs C, Garber JE, Neuhausen SL, Matloff E, Eeles R, Pichert G, Van t’veer L, Tung N, Weitzel JN, Couch FJ, Rubinstein WS, Ganz PA, Daly MB, Olopade OI, Tomlinson G, Schildkraut J, Blum JL, Rebbeck TR. Association of Risk-Reducing Surgery in BRCA1 or BRCA2 Mutation Carriers with Cancer Risk and Mortality. JAMA. 2010;304(9):967-975. doi:10.1001/jama.2010.1237
- Finch AP, Lubinski J, Møller P, Singer CF, Karlan B, Senter L, Rosen B, Maehle L, Ghadirian P, Cybulski C, Huzarski T, Eisen A, Foulkes WD, Kim-Sing C, Ainsworth P, Tung N, Lynch HT, Neuhausen S, Metcalfe KA, Thompson I, Murphy J, Sun P, Narod SA. Impact of Oophorectomy on Cancer Incidence and Mortality in Women with a BRCA1 or BRCA2 Mutation. Journal of Clinical Oncology.2014;32(15):1547-1553. doi:10.1200/JCO.2013.53.2820
- Hartmann LC, Schaid DJ, Woods JE, Crotty TP, Myers JL, Arnold PG, Petty PM, Sellers TA, Johnson JL, McDonnell SK, Frost MH, Jenkins RB. Efficacy of Bilateral Prophylactic Mastectomy in Women with a Family History of Breast Cancer. New England Journal of Medicine. 1999;340(2):77-84. doi: 10.1056/NEJM199901143400201
- Hartmann LC, Lindor NM. The Role of Risk-Reducing Surgery in Hereditary Breast and Ovarian Cancer. New England Journal of Medicine. 2016;374(5):454-468. doi:10.1056/NEJMra1503523
- Kotsopoulos J, Gronwald J, Karlan B, Rosen B, Huzarski T, Moller P, Lynch HT, Singer CF, Senter L, Neuhausen SL, Tung N, Eisen A, Foulkes WD, Ainsworth P, Sun P, Lubinski J, Narod SA, Hereditary Ovarian Cancer Clinical Study Group. Age-Specific Ovarian Cancer Risks among Women with a BRCA1 or BRCA2 Mutation. Gynecologic Oncology. 2018;150(1):85-91. doi:10.1016/j.ygyno.2018.05.011
- Kuchenbaecker KB, Hopper JL, Barnes DR, Phillips KA, Mooij TM, Roos-Blom MJ, Jervis S, van Leeuwen FE, Milne RL, Andrieu N, Goldgar DE, Terry MB, Rookus MA, Easton DF, Antoniou AC, BRCA1 and BRCA2 Cohort Consortium. Risks of Breast, Ovarian, and Contralateral Breast Cancer for BRCA1 and BRCA2 Mutation Carriers. JAMA. 2017;317(23):2402-2416. doi:10.1001/jama.2017.7112
- Mavaddat N et al. Pathology of Breast and Ovarian Cancers among BRCA1 and BRCA2 Mutation Carriers: Results from the Consortium of Investigators of Modifiers of BRCA1/2 (CIMBA). Cancer Epidemiology, Biomarkers & Prevention. 2012;21(1):134-147. doi:10.1158/1055-9965.EPI-11-0775
- Rebbeck TR, Friebel T, Lynch HT, Neuhausen SL, van ‘t Veer L, Garber JE, Evans GR, Narod SA, Isaacs C, Matloff E, Daly MB, Olopade OI, Weber BL. Bilateral Prophylactic Mastectomy Reduces Breast Cancer Risk in BRCA1 and BRCA2 Mutation Carriers: The PROSE Study Group. Journal of Clinical Oncology. 2004;22(6):1055-1062. doi:10.1200/JCO.2004.04.188
- Rebbeck TR, Mitra N, Wan F, Sinilnikova OM, Healey S, McGuffog L, Mazoyer S, Chenevix-Trench G, Easton DF, Antoniou AC, Nathanson KL, CIMBA Consortium. Association of Type and Location of BRCA1 and BRCA2 Mutations with Risk of Breast and Ovarian Cancer. JAMA. 2015;313(13):1347-1361. doi:10.1001/jama.2014.5985
- Antoniou AC et al. Reproductive and hormonal factors, and ovarian cancer risk for BRCA1 and BRCA2 mutation carriers: results from the International BRCA1/2 Carrier Cohort Study. Cancer Epidemiology and Prevention Biomarkers. 2009;18(2):601-610. doi:10.1158/1055-9965.EPI-08-0546
- Rice MS, Murphy MA, Tworoger SS. Tubal ligation, hysterectomy and ovarian cancer: a meta-analysis. Journal of Ovarian Research. 2012;5(13):1-16. doi:10.1186/1757-2215-5-13